|
How many GRAMS of boron tribromide are present in 3. 0:00 / 1:01 How to Write the Formula for Chromium (II) iodide Wayne Breslyn 622K subscribers Subscribe 1.4K views 1 year ago In this video we'll write the correct formula for Chromium (II). It is a black solid that is used to prepare other. How many MOLES of boron tribromide are present in 2.23 grams of this compound? moles 2. Chromium(III) iodide, also known as chromium triiodide, is an inorganic compound with the formula CrI3. The dissociation will be complete and irreversible. The conduction will happen because of the presence of the ions. KNO2 - Potassium nitrite SF6 - Sulfur hexafluoride CrPO4 - Chromium(III) phosphate (There is no compoun like Cr(PO2)2 ) NiCO3 - Nickel(II) carbonate (There is no compound like NiCO2 ) CCl4 - Carbon tetrachloride. Answer: Cr (NO) (s) Cr (aq) + NO (aq) Explanation: A strong electrolyte is a substance that dissolves in water, dissociating in its ions, and making the solution an electric conductor. This can be represented as, CrCl2 (aq) + 2NaOH (aq) Cr (OH)2 (s) + 2NaCl (aq) This is th. chromium (II) iodide chromium (III) chloride cobalt (II) bromide copper (II) oxide. References Use the Red Use the References to access important values if needed for th 1. SOLUTION:- a) Aqueous chromium (II) chloride on reaction with aqueous sodium hydroxide gives solid chromium (II) hydroxide and aqueous sodium chloride. REVIEW TOPICS Relerences Use the References to access important values if needed for this questi For the molecular compound nitrogen monoxide, what would you multiply "grams of NO" by to get the units "moles of NO" ? (number) (unit) grams NO = moles NO (number) (unit) Submit Answer Retry Entire Group 4 more group attempts remaining 
Use the References to access important values if needed for this que For the molecular compound phosphorus trichloride, what would you multiply "moles of PCI " by to get the units "grams of PC13" ? (number) (unit) moles PC1z = grams PC13 (number) (unit) In progress L Answer Retry Entire Group 4 more group attempts remaining 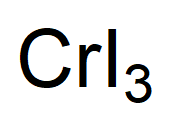
The net ionic equation for this reaction is: 3c. Consider the reaction when aqueous solutions of zinc sulfate and iron (III) acetate are combined. The net ionic equation for this reaction is: 3b. 
Formula Molar mass of ion g/mol Number of ions mol Mass of ion in one mole of chromium(II) iodide 8 Cation = Anion g/mol mol Molar mass chromium(IT) iodide g/mol Submit Answer Retry Entire Group 4 more group attempts remaining Consider the reaction when aqueous solutions of chromium (II) nitrate and barium iodide are combined. Complete the table below for calculating the molar mass of the ionic compound chromium(II) iodide.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
